|
As the nucleus of the atom is at the center thus a proton holding a positive charge is present at the center of the atomic structure. It’s a crucial component of an atom that forms the nucleus of an atom with the neutron. Proton is another major particle of an atom with a charge of positive polarity. 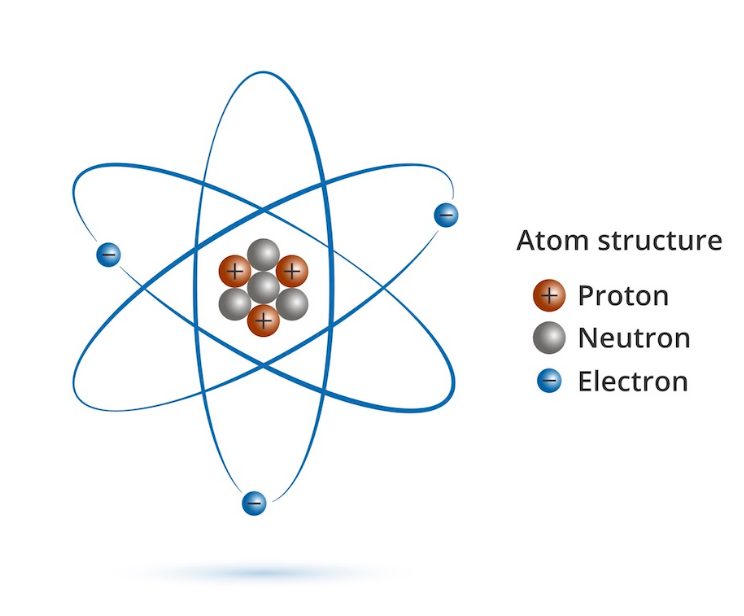
And all the electrons are considered to be identically similar to each other. The electron charge is measured in Coulomb and has a value of about 1.602 * 10 -19 C. And an electron is considered to have a unit electrical charge generally represented as e. In the case of conductors, the movement of electrons is the reason for the flow of current. While when external energy is supplied to the electrons then they move from an atom to another.īasically, the energy supplied to the electrons frees them from the shells thus they become mobile and get attached to its nearby atom whenever there is a deficiency of electron in that particular atom. Inside an atom, ideally, electrons are present in spherical shells and move around the nucleus in an orbital path. These are very difficult to get added or removed.Ī subatomic particle of an atom that holds an electric charge of negative polarity. 
These can be easily removed from or added to an atom. Outside the nucleus in a well-defined orbit. Therefore, has various differentiating factors which we will discuss here. Among electron, proton, and neutron, the electrons and protons are negatively and positively charged respectively while the neutrons are neutrally charged particles.Įlectrons and protons hold different properties and are present at different locations inside the atom. The electric charge of the atom maintains the bond between the atoms in a molecule. Multiple atoms form a molecule and the atoms inside a molecule are attached by chemical bonds. While this atom itself has 3 major subatomic particles which are known as electron, proton, and neutron. Treating the charges on nitrogen and argon as the sums of charges on protons, electrons and neutrons, it is deduced that the proton charge is (1±4×10 −20) e and the charge on the neutron is less than 4 × −20 e.We know that an atom is regarded as the smallest particle as it is the fundamental unit by which a matter is composed.  
We have found that the charge on an argon atom (18 protons, 18 electrons and 22 neutrons) is not greater than 8×10 −20 e and that on a nitrogen molecule (14 protons, 14 electrons and 14 neutrons) is not greater than 12 × 10 −20 e. It therefore seemed desirable to find whether matter in which there is an excess of neutrons is electrically neutral. Since matter also contains neutrons, they have assumed in effect that the neutron has a charge equal to that of a hydrogen atom but the neutron might equally well have a charge opposite to that of a hydrogen atom, in which case their experiment does not settle the point at issue, since carbon dioxide contains equal numbers of protons and neutrons (to within 0.1 per cent). In fact, Piccard and Kessler 3 attempted to detect such a difference in 1925, and found that a molecule of carbon dioxide did not have an electric charge greater than 2×10 −19 e, where - e is the electronic charge, from which they concluded that the magnitudes of the proton and electron charges were the same to within 5 parts in 10 21, assuming that matter was built entirely of protons and electrons. IT has recently been suggested by Bondi and Ly ttleton 1,2 that the magnitudes of the electric charges on the proton and electron may differ by a little more than one part in 10 18, in which case electrostatic forces would cause the universe to expand.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
